Yes we know..... you think lancet or Harvard medical schools ID and epidemiology chiefs don’t know about double blind studies. Not all of medicine is based of double blind RCTs. There are so many things we do that we know work but are not based off double blind RCTs. You can’t just ignore all the evidence in favour of one trial. Depending on the effect size even a double blind RCT may not be powered to make conclusions beyond its sample or make statistically significant conclusions. But yes double blind RCT are level one evidence but again you can’t ignore other studies just cause their not double blinded....
I like how people are being extremely critical of a well designed study that shows no efficacy of hcq yet they support YouTube videos or a single extremely poorly designed French study that fails to explain how some participants were magically/conveniently disappeared to follow up. If we are going to be this critical let’s be this critical towards all the evidence. Doing so would automatically disqualify 100% of the things your YouTube guy says. Lol
I m sure that all doctor and clinical trial are aware of random double blinded studies, espically medical school, but all I hear is excuses and excuses ! What is so difficult making clinical doctor and patient not knowing that they are giving a real drug or a placebo is to keep everyone honest!
Just like WHO telling everyone the public mask or Health Canada are not useful to public, not to wear mask or NO human to Human transmission inJanuary!
The public is not stupid ! Anyone can go to YouTube to learn how to wear a mask probably or if the governments is so concerned that people wearing a mask wrong. They can advertise on NEWS or TV showing the public how to wear a Mask properly!
Getting back to double blinded studies topics!
Very easy to have a double blinded studies, guess in USA everything is driven by profit! Double blinded studies prevent confirmation biases!
Double blinded studies conflict of interest and prevent favouring one drug over other( profit motivations may have some share in that trial easy to make a quick buck " pump & dump" for a quick profits!
That the Gold Standard! That is to prevent conflict of interest! That extremely important!
Example your the VA Hospital had two studies one HCQ not randomly controlled trial poorly designed and HCQ trial by the same hospital that was conducting Remsivar which was better designed trial . That was obviously showed a conflict of interest ( VA Hospital administration receives grant or money from Glead Health maker of remsivar) and then they touted how wonderful Reimsivar !
Even a preception of Conflict of interest is a NO NO !
Politics in medicine and confirmation biased in any RCT trial need to be questioned! Confirmation biased also led to promoting one drug over another due to profit motivations also! The clinical doctor could also have share in remisvidar !
So double blinded studies put everyone in a equal footing or put everyone in the same level playing field!
Here is the result of the remdiesver trial that was touted by Dr Fauci
The Remdesivir Study Is Finally Out: Drug Only Helped Those On Oxygen, Finds Mortality Too High For Standalone Treatment
Remember when the market soared on several days in April on the Facui-touted Remdesivir study which, according to StatNews and various other unofficial sources of rumors, was a smashing success only for the optimism to fizzle as many questions emerged, and as the Gilead drug quietly faded from the public's consciousness and was replaced by various coronavirus vaccine candidates such as those made by the greatly hyped Moderna (whose insiders https://investors.modernatx.com/static-files/03ba600f-801d-4609-a7fb-332ac507f76f
just can't stop selling company stock). https://www.cnn.com/2020/05/22/investing/moderns-coronavirus-vaccine-stock-sales/index.html
Adam Feuerstein
@adamfeuerstein
https://twitter.com/adamfeuerstein/status/1263958445646065664
Friday 6 pm. Fking ridiculous.
Quote Tweet
... According to a pivotal study published in the New England Journal of Medicine late on Friday,
https://www.nejm.org/doi/full/10.1056/NEJMoa2007764
Remdesivir, which was authorized to treat Covid-19 in a group of 1063 adults and children (split into two groups, one receiving placebo instead of remdesivir) who need i) supplemental oxygen, ii) a ventilator or iii) extracorporeal membrane oxygenation (ECMO), only significantly helped those on supplemental oxygen.
Meanwhile, and explaining the 6pm release on a Friday, the study also found no marked benefit from remdesivir for those who were healthier and didn’t need oxygen or those who were sicker, requiring a ventilator or a heart-lung bypass machine.
The NEJM, almost apologetically, stated that "the lack of benefit seen in the other groups might have stemmed from a smaller number of patients in each group."
Still, as a result of the partial benefit for patients in the supplemental oxygen group, the study from the National Institute of Allergy and Infectious Diseases was evaluated early and led to the authorization of remdesivir before the full trial was completed.
Our findings highlight the need to identify Covid-19 cases and start antiviral treatment before the pulmonary disease progresses to require mechanical ventilation.
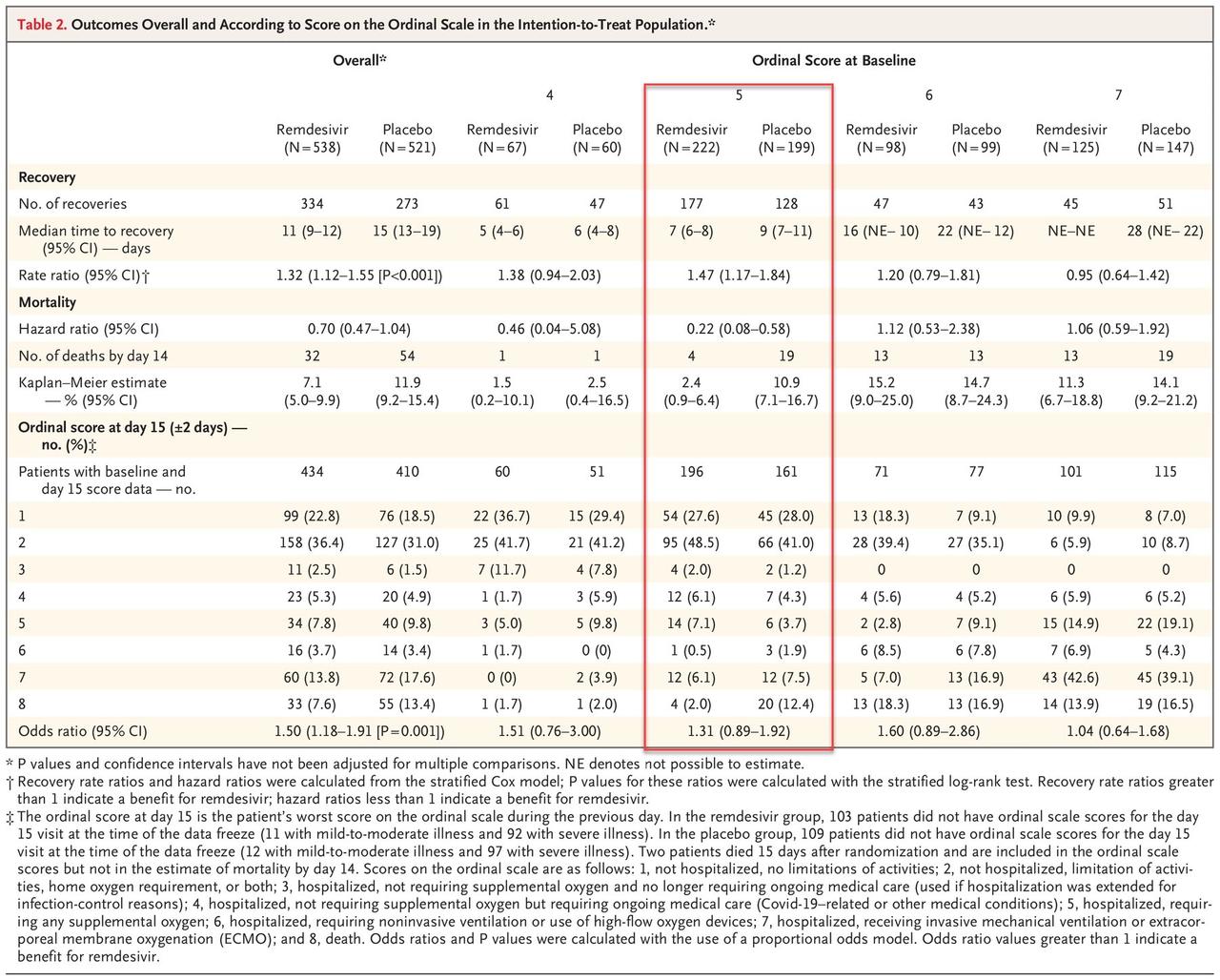
Some more details on the study, which was a "rank test of the time to recovery with remdesivir as compared with placebo, with stratification by disease severity":
The primary outcome measure was the time to recovery, defined as the first day, during the 28 days after enrollment, on which a patient satisfied categories 1, 2, or 3 on the eight-category ordinal scale. The categories are as follows:
1. not hospitalized, no limitations of activities;
2. not hospitalized, limitation of activities, home oxygen requirement, or both;
3. hospitalized, not requiring supplemental oxygen and no longer requiring ongoing medical care (used if hospitalization was extended for infection-control reasons);
4. hospitalized, not requiring supplemental oxygen but requiring ongoing medical care (Covid-19–related or other medical conditions);
5. 5, hospitalized, requiring any supplemental oxygen;
hospitalized, requiring noninvasive ventilation or use of high-flow oxygen devices;
hospitalized, receiving invasive mechanical ventilation or extracorporeal membrane oxygenation (ECMO); and death.
The results are summarized below, highlighting the only group that showed a statistically significant improvement in outcomes as a result of taking the drug vs placebo.
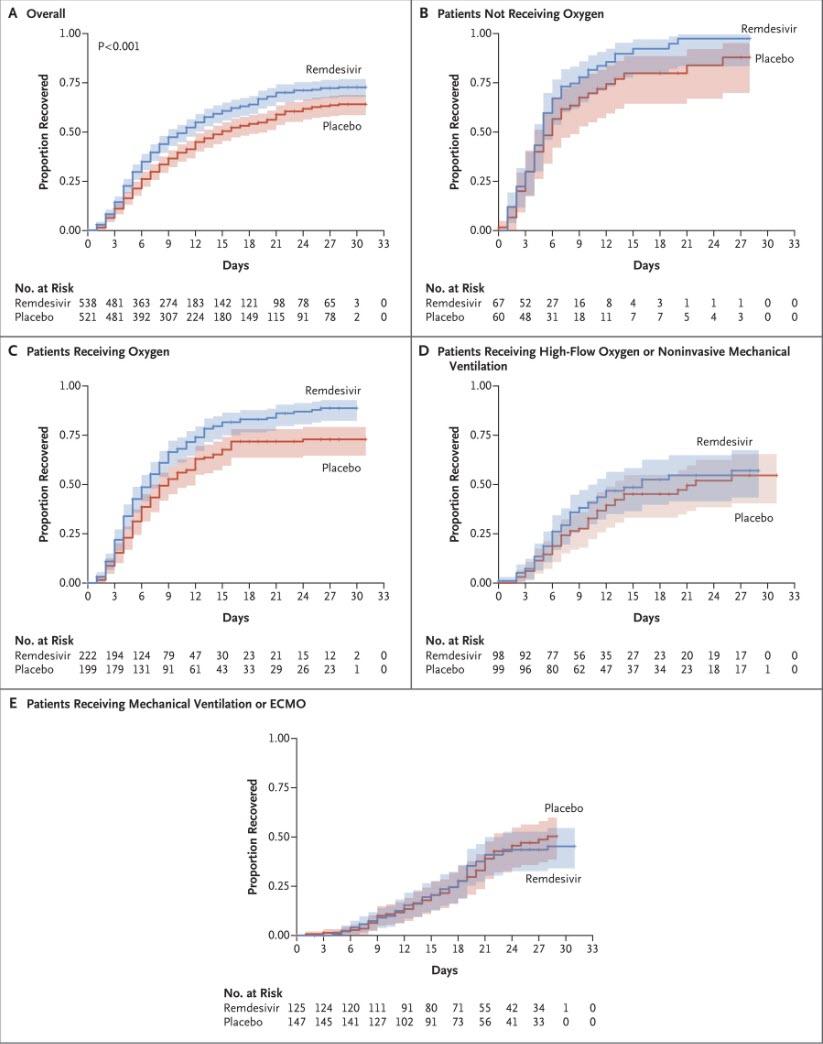
A visual representation of the outcomes is below; it shows that whereas there was a modest benefit only to patients who were receiving oxygen, the results were statistically insignificant vs placebo for patients not receiving oxygen, while in a surprising twist patients on high-flow oxygen or mechanical ventilator/ECMO did modestly better in the placebo group than those taking remdesivir. Also, the overall results showed a very modest, but not statistically significant improvement in the remdesivir group vs placebo (box A).
Another disappointment: the study found that overall "mortality was numerically lower in the remdesivir group than in the placebo group, but the difference was not significant", in other words the alleged "miracle drug" has largely the same effect as a placebo in terms of overall disease mortality.
The study authors also note that the "findings in our trial should be compared with those observed in a randomized trial from China in which 237 patients were enrolled (158 assigned to remdesivir and 79 to placebo).... That trial failed to complete full enrollment (owing to the end of the outbreak), had lower power than the present trial (owing to the smaller sample size and a 2:1 randomization), and was unable to demonstrate any statistically significant clinical benefits of remdesivir."
Finally, the study found that while mortality was modestly lower for the remdesivir arm, it was not significantly so, at 7.1% at 14 days on drug versus 11.9% on placebo.
In conclusion, while the "preliminary findings support the use of remdesivir for patients who are hospitalized with Covid-19 and require supplemental oxygen therapy" the study goes on to warn that "given high mortality despite the use of remdesivir, it is clear that treatment with an antiviral drug alone is not likely to be sufficient."
The study's recommendation:
Future strategies should evaluate antiviral agents in combination with other therapeutic approaches or combinations of antiviral agents to continue to improve patient outcomes in Covid-19.
So a generally disappointing outcome, one which would lead to a drop in the market. Nonsense: think of all the spin, and why this is in fact great news for stocks: Remdesivir may be a dud as a "silver bullet" to curing covid, leading to statistically significant improvement in only a very limited subset of infected patients and "high mortality" for those taking it, but at least the algos will have a whole lot of other "miracle drugs" to levitate them as optimism that the next remdesivir is just around the corner. In short: rinse, rumor, and repeat... and then save the bad news for 6pm on a Friday.
Oh, and for those asking about the "official" reason why the NE Journal of Medicine waited until just the right time to make sure nobody reads the results, here it is:
Adam Feuerstein @adamfeuerstein
@adamfeuerstein
I asked NEJM spox to explain the Friday 6 pm release of the remdesivir study. Her response is below.
— Adam Feuerstein (@adamfeuerstein) May 22, 2020
https://twitter.com/adamfeuerstein/...-helped-those-oxygen-finds-mortality-too-high
RE: remdesivir study released:
Adam,
We received manuscripts recently and have been
working with the authors to get it published as rapidly as
possible. Editorial was working until moments before publication
to finished it. We didn't want to wait until Memorial Weekend to
pass before publishing.
JEN
The full study is available below here.
https://www.nejm.org/doi/full/10.1056/NEJMoa2007764
Even by your own posting you I guesssing you show sort a biased against right wings calling them as alt right ( That my initial perception but I can be wrong on that. If I will apologize if I guess wrong,) .
A double blinded studies will put any arguments to rest once and for all ! So time will not be wasted or
but you liberals doctors will or not deliberately will not designed that that of clinical trial!
Is a sad fact today due to politics life will can be saved to a small subset patients that may benefit !
Finding some sort of excuses for not designing a double blinded studies is reek of politics or confirmation biased ! We know unfortunately medicine have been politicized that includes you!
Even my your own
admission from your previous posting above about 5% of Facebook polling by medical doctor.That 5% of medical doctor do that HCQ as a preventative medicine against covid19.
Answers these questions. Honestly answer these questions ! Do you have a HCQ prescription for any of your family members or close friends? Do you have your own supply of HCQ?
Are you presently taking HCQ as a preventative medicine against Covid19?
If you for some reason at the.first symptoms of covid19 ( example losing sense of small or taste) or a sign of flu symptoms would you considered taking HCQ with zpak and Zinc.
Would you even consider taking HCQ?
And do you know any of your medical colleagues presently taking HCQ?
Would you consider taking HCQ?
Do you know any of your medical colleagues that confided in you about they would consider taking HCQ if there aren't any better drugs that are available or vaccines available ?
And why are doctor hoarding HCQ?
https://www.propublica.org/article/...escriptions-for-themselves-and-their-families
Answer above questions honestly!
Do your hypocrate oath mean anything to you?
Remember your hypocrate oath!
If you can look yourself in the mirror after answering this truthfully questions I posed then your doctor so called expert opinions will hold more weight to me!! I reserved the right to change my mind if the new data , hopefully double blinded studies come out disproving all naysayers of HCQ or those suffer TDS ( Hate anything from the right / conservative view point). I will only follow or believe in randomly double blinded studies, because it the Gold Standard. Any thing less in my opinion is just excuse & excuse! Random Double blinded studied or not expensive probably the same cost as a random controlled trial. I can see no extra cost involvement of not letting the patients or doctor that they are giving a placebo or a drug/ vaccine!
Majority of medical Doctors have integrity and Honesty in their moral value but you do a few bad apple example (WHO , others) !
So Again Please answer the above questions I posted to you!
!